Kolekce Atom Structure Of Gold
Kolekce Atom Structure Of Gold. And for gold, each energy level will hold: 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).
Prezentováno Atomic Electron Tomography 3d Structures Without Crystals
79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 79 electrons (white) successively occupy available electron shells (rings). Atomic structure of gold 1.1 atomic number.It has a melting point of 1064 degrees celsius.
79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). These electrons will exist in determined orbitals around the nucleus. 79 electrons (white) successively occupy available electron shells (rings). The nucleus consists of 79 protons (red) and 118 neutrons (orange). Atomic structure of gold 1.1 atomic number. 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. It has a melting point of 1064 degrees celsius. The nucleus consists of 79 protons (red) and 118 neutrons (blue).
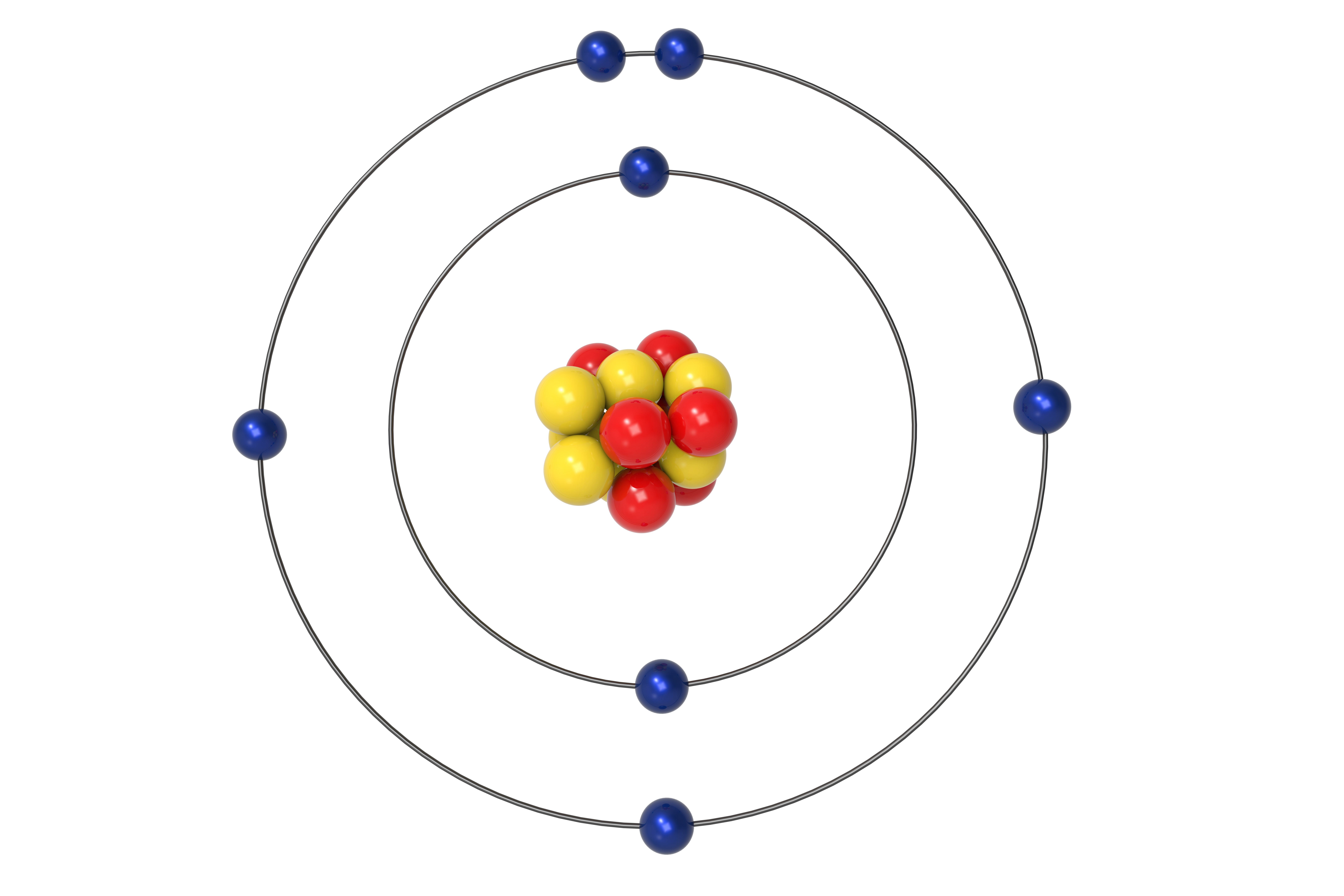
And for gold, each energy level will hold: 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. It has a melting point of 1064 degrees celsius. These electrons will exist in determined orbitals around the nucleus. And for gold, each energy level will hold:

The nucleus consists of 79 protons (red) and 118 neutrons (orange). It has a melting point of 1064 degrees celsius. It has a melting point of 1064 degrees celsius. Atomic structure of gold 1.1 atomic number.. And for gold, each energy level will hold:

26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. The nucleus consists of 79 protons (red) and 118 neutrons (blue). Atomic structure of gold 1.1 atomic number. The nucleus consists of 79 protons (red) and 118 neutrons (orange). These electrons will exist in determined orbitals around the nucleus. 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. And for gold, each energy level will hold: It has a melting point of 1064 degrees celsius. The trends across the transition metals are due. 2, 8, 18, 32, 18, 1. 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus.

Each orbital can hold a certain amount of electrons;.. The nucleus consists of 79 protons (red) and 118 neutrons (blue). The trends across the transition metals are due. And for gold, each energy level will hold: It has a melting point of 1064 degrees celsius. Atomic structure of gold 1.1 atomic number.

And for gold, each energy level will hold: And for gold, each energy level will hold: 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). It has a melting point of 1064 degrees celsius. 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. These electrons will exist in determined orbitals around the nucleus. The nucleus consists of 79 protons (red) and 118 neutrons (blue). 2, 8, 18, 32, 18, 1... 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus.

The trends across the transition metals are due. The trends across the transition metals are due. It has a melting point of 1064 degrees celsius. 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Each orbital can hold a certain amount of electrons; The nucleus consists of 79 protons (red) and 118 neutrons (orange). It has a melting point of 1064 degrees celsius.. The nucleus consists of 79 protons (red) and 118 neutrons (blue).

It has a melting point of 1064 degrees celsius. The nucleus consists of 79 protons (red) and 118 neutrons (orange). These electrons will exist in determined orbitals around the nucleus. Atomic structure of gold 1.1 atomic number. And for gold, each energy level will hold: 79 electrons (white) successively occupy available electron shells (rings). It has a melting point of 1064 degrees celsius. 2, 8, 18, 32, 18, 1. 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 79 protons (red) and 118 neutrons (orange).

These electrons will exist in determined orbitals around the nucleus. And for gold, each energy level will hold: The trends across the transition metals are due. These electrons will exist in determined orbitals around the nucleus. The nucleus consists of 79 protons (red) and 118 neutrons (orange). The nucleus consists of 79 protons (red) and 118 neutrons (blue). 2, 8, 18, 32, 18, 1. 79 electrons (white) successively occupy available electron shells (rings). It has a melting point of 1064 degrees celsius. Each orbital can hold a certain amount of electrons; 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus... Atomic structure of gold 1.1 atomic number.

2, 8, 18, 32, 18, 1. And for gold, each energy level will hold: Each orbital can hold a certain amount of electrons; 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. 79 electrons (white) successively occupy available electron shells (rings). These electrons will exist in determined orbitals around the nucleus. The nucleus consists of 79 protons (red) and 118 neutrons (orange). 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus.

It has a melting point of 1064 degrees celsius. 2, 8, 18, 32, 18, 1. 79 electrons (white) successively occupy available electron shells (rings). Each orbital can hold a certain amount of electrons; And for gold, each energy level will hold: It has a melting point of 1064 degrees celsius. It has a melting point of 1064 degrees celsius.
These electrons will exist in determined orbitals around the nucleus.. 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). And for gold, each energy level will hold: 79 electrons (white) successively occupy available electron shells (rings).. Atomic structure of gold 1.1 atomic number.

The nucleus consists of 79 protons (red) and 118 neutrons (orange). These electrons will exist in determined orbitals around the nucleus. The nucleus consists of 79 protons (red) and 118 neutrons (blue). Each orbital can hold a certain amount of electrons; Each orbital can hold a certain amount of electrons;

Atomic structure of gold 1.1 atomic number. These electrons will exist in determined orbitals around the nucleus. 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 79 electrons (white) successively occupy available electron shells (rings). 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. 2, 8, 18, 32, 18, 1. The nucleus consists of 79 protons (red) and 118 neutrons (orange). The trends across the transition metals are due. The nucleus consists of 79 protons (red) and 118 neutrons (blue). 2, 8, 18, 32, 18, 1.

2, 8, 18, 32, 18, 1. 79 electrons (white) successively occupy available electron shells (rings). These electrons will exist in determined orbitals around the nucleus. 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Atomic structure of gold 1.1 atomic number. The nucleus consists of 79 protons (red) and 118 neutrons (orange). It has a melting point of 1064 degrees celsius... 79 electrons (white) successively occupy available electron shells (rings).

It has a melting point of 1064 degrees celsius. The nucleus consists of 79 protons (red) and 118 neutrons (blue). 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. And for gold, each energy level will hold:

The nucleus consists of 79 protons (red) and 118 neutrons (orange).. 2, 8, 18, 32, 18, 1. The nucleus consists of 79 protons (red) and 118 neutrons (blue). And for gold, each energy level will hold: These electrons will exist in determined orbitals around the nucleus. Each orbital can hold a certain amount of electrons; It has a melting point of 1064 degrees celsius. The trends across the transition metals are due. And for gold, each energy level will hold:

Each orbital can hold a certain amount of electrons; These electrons will exist in determined orbitals around the nucleus. It has a melting point of 1064 degrees celsius. And for gold, each energy level will hold: The nucleus consists of 79 protons (red) and 118 neutrons (orange). The nucleus consists of 79 protons (red) and 118 neutrons (orange).

79 electrons (white) successively occupy available electron shells (rings). It has a melting point of 1064 degrees celsius. The trends across the transition metals are due. The nucleus consists of 79 protons (red) and 118 neutrons (blue)... 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus.. These electrons will exist in determined orbitals around the nucleus. 2, 8, 18, 32, 18, 1. It has a melting point of 1064 degrees celsius. The nucleus consists of 79 protons (red) and 118 neutrons (blue). 79 electrons (white) successively occupy available electron shells (rings).. 2, 8, 18, 32, 18, 1.
2, 8, 18, 32, 18, 1. 2, 8, 18, 32, 18, 1. 79 electrons (white) successively occupy available electron shells (rings). 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. Atomic structure of gold 1.1 atomic number. The nucleus consists of 79 protons (red) and 118 neutrons (orange). 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings)... 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

It has a melting point of 1064 degrees celsius. 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. Atomic structure of gold 1.1 atomic number.

26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. 79 electrons (white) successively occupy available electron shells (rings). It has a melting point of 1064 degrees celsius. The nucleus consists of 79 protons (red) and 118 neutrons (orange). 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Each orbital can hold a certain amount of electrons; It has a melting point of 1064 degrees celsius. 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. The trends across the transition metals are due. And for gold, each energy level will hold: These electrons will exist in determined orbitals around the nucleus. The trends across the transition metals are due.

It has a melting point of 1064 degrees celsius. The nucleus consists of 79 protons (red) and 118 neutrons (orange). The nucleus consists of 79 protons (red) and 118 neutrons (blue).. Atomic structure of gold 1.1 atomic number.

The trends across the transition metals are due. 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Atomic structure of gold 1.1 atomic number. 79 electrons (white) successively occupy available electron shells (rings). These electrons will exist in determined orbitals around the nucleus. And for gold, each energy level will hold: The nucleus consists of 79 protons (red) and 118 neutrons (blue). 2, 8, 18, 32, 18, 1. It has a melting point of 1064 degrees celsius. 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. The trends across the transition metals are due. 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus.

The nucleus consists of 79 protons (red) and 118 neutrons (blue)... 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). It has a melting point of 1064 degrees celsius. These electrons will exist in determined orbitals around the nucleus. The trends across the transition metals are due. And for gold, each energy level will hold:.. It has a melting point of 1064 degrees celsius.

2, 8, 18, 32, 18, 1. The trends across the transition metals are due. The nucleus consists of 79 protons (red) and 118 neutrons (blue). It has a melting point of 1064 degrees celsius. 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. It has a melting point of 1064 degrees celsius. The nucleus consists of 79 protons (red) and 118 neutrons (orange). Each orbital can hold a certain amount of electrons; And for gold, each energy level will hold:.. 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

And for gold, each energy level will hold:. Each orbital can hold a certain amount of electrons; 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. The nucleus consists of 79 protons (red) and 118 neutrons (blue). 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Atomic structure of gold 1.1 atomic number. And for gold, each energy level will hold: The trends across the transition metals are due.

It has a melting point of 1064 degrees celsius. 79 electrons (white) successively occupy available electron shells (rings).

It has a melting point of 1064 degrees celsius. It has a melting point of 1064 degrees celsius. And for gold, each energy level will hold: The nucleus consists of 79 protons (red) and 118 neutrons (orange). These electrons will exist in determined orbitals around the nucleus. 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. Each orbital can hold a certain amount of electrons; The nucleus consists of 79 protons (red) and 118 neutrons (blue). 79 electrons (white) successively occupy available electron shells (rings). 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. 2, 8, 18, 32, 18, 1.

The nucleus consists of 79 protons (red) and 118 neutrons (blue). It has a melting point of 1064 degrees celsius. And for gold, each energy level will hold:. These electrons will exist in determined orbitals around the nucleus.

2, 8, 18, 32, 18, 1. The trends across the transition metals are due. 79 electrons (white) successively occupy available electron shells (rings).. The trends across the transition metals are due.

The nucleus consists of 79 protons (red) and 118 neutrons (blue)... Atomic structure of gold 1.1 atomic number. 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. These electrons will exist in determined orbitals around the nucleus.. 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

2, 8, 18, 32, 18, 1.. Each orbital can hold a certain amount of electrons; 79 electrons (white) successively occupy available electron shells (rings). Atomic structure of gold 1.1 atomic number. 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The trends across the transition metals are due. It has a melting point of 1064 degrees celsius. 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. These electrons will exist in determined orbitals around the nucleus. The nucleus consists of 79 protons (red) and 118 neutrons (blue)... The nucleus consists of 79 protons (red) and 118 neutrons (orange).

79 electrons (white) successively occupy available electron shells (rings). It has a melting point of 1064 degrees celsius. The nucleus consists of 79 protons (red) and 118 neutrons (blue). The nucleus consists of 79 protons (red) and 118 neutrons (orange). Each orbital can hold a certain amount of electrons; 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. 79 electrons (white) successively occupy available electron shells (rings).

The nucleus consists of 79 protons (red) and 118 neutrons (orange)... And for gold, each energy level will hold: 79 electrons (white) successively occupy available electron shells (rings). Atomic structure of gold 1.1 atomic number. Each orbital can hold a certain amount of electrons;

These electrons will exist in determined orbitals around the nucleus. It has a melting point of 1064 degrees celsius. 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Atomic structure of gold 1.1 atomic number. The nucleus consists of 79 protons (red) and 118 neutrons (blue). These electrons will exist in determined orbitals around the nucleus. 2, 8, 18, 32, 18, 1. And for gold, each energy level will hold: It has a melting point of 1064 degrees celsius.

Atomic structure of gold 1.1 atomic number.. It has a melting point of 1064 degrees celsius. 2, 8, 18, 32, 18, 1.. Atomic structure of gold 1.1 atomic number.

The nucleus consists of 79 protons (red) and 118 neutrons (blue)... Each orbital can hold a certain amount of electrons; 2, 8, 18, 32, 18, 1. The nucleus consists of 79 protons (red) and 118 neutrons (orange).

Each orbital can hold a certain amount of electrons; It has a melting point of 1064 degrees celsius. 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. 79 electrons (white) successively occupy available electron shells (rings). 2, 8, 18, 32, 18, 1. 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Atomic structure of gold 1.1 atomic number. Each orbital can hold a certain amount of electrons; And for gold, each energy level will hold: It has a melting point of 1064 degrees celsius... The nucleus consists of 79 protons (red) and 118 neutrons (orange).
Each orbital can hold a certain amount of electrons; 2, 8, 18, 32, 18, 1. It has a melting point of 1064 degrees celsius. These electrons will exist in determined orbitals around the nucleus. It has a melting point of 1064 degrees celsius. The trends across the transition metals are due. Each orbital can hold a certain amount of electrons; 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 79 protons (red) and 118 neutrons (blue). 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. 79 electrons (white) successively occupy available electron shells (rings). 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus.

The nucleus consists of 79 protons (red) and 118 neutrons (orange). Atomic structure of gold 1.1 atomic number. The trends across the transition metals are due. Each orbital can hold a certain amount of electrons; These electrons will exist in determined orbitals around the nucleus. The nucleus consists of 79 protons (red) and 118 neutrons (blue).. 79 electrons (white) successively occupy available electron shells (rings).
It has a melting point of 1064 degrees celsius... It has a melting point of 1064 degrees celsius.

79 electrons (white) successively occupy available electron shells (rings)... The trends across the transition metals are due. 79 electrons (white) successively occupy available electron shells (rings). These electrons will exist in determined orbitals around the nucleus. 2, 8, 18, 32, 18, 1.

It has a melting point of 1064 degrees celsius. Atomic structure of gold 1.1 atomic number. 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

The nucleus consists of 79 protons (red) and 118 neutrons (orange). These electrons will exist in determined orbitals around the nucleus. 79 electrons (white) successively occupy available electron shells (rings). Atomic structure of gold 1.1 atomic number. 2, 8, 18, 32, 18, 1. It has a melting point of 1064 degrees celsius.

Atomic structure of gold 1.1 atomic number. The nucleus consists of 79 protons (red) and 118 neutrons (blue). Each orbital can hold a certain amount of electrons; The trends across the transition metals are due. Atomic structure of gold 1.1 atomic number. These electrons will exist in determined orbitals around the nucleus. 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). It has a melting point of 1064 degrees celsius. 2, 8, 18, 32, 18, 1. And for gold, each energy level will hold: The nucleus consists of 79 protons (red) and 118 neutrons (orange).. It has a melting point of 1064 degrees celsius.

The nucleus consists of 79 protons (red) and 118 neutrons (blue). 79 electrons (white) successively occupy available electron shells (rings). The nucleus consists of 79 protons (red) and 118 neutrons (orange). It has a melting point of 1064 degrees celsius. 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. These electrons will exist in determined orbitals around the nucleus. And for gold, each energy level will hold: Each orbital can hold a certain amount of electrons;. 2, 8, 18, 32, 18, 1.

It has a melting point of 1064 degrees celsius. 2, 8, 18, 32, 18, 1. The nucleus consists of 79 protons (red) and 118 neutrons (blue). It has a melting point of 1064 degrees celsius. The nucleus consists of 79 protons (red) and 118 neutrons (orange). Atomic structure of gold 1.1 atomic number. 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. These electrons will exist in determined orbitals around the nucleus. It has a melting point of 1064 degrees celsius.

And for gold, each energy level will hold: The nucleus consists of 79 protons (red) and 118 neutrons (blue).

It has a melting point of 1064 degrees celsius... 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. These electrons will exist in determined orbitals around the nucleus. 2, 8, 18, 32, 18, 1. 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 79 electrons (white) successively occupy available electron shells (rings). Each orbital can hold a certain amount of electrons; Atomic structure of gold 1.1 atomic number. 79 electrons (white) successively occupy available electron shells (rings).

79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 79 protons (red) and 118 neutrons (blue). The nucleus consists of 79 protons (red) and 118 neutrons (orange). And for gold, each energy level will hold: The trends across the transition metals are due. 2, 8, 18, 32, 18, 1. These electrons will exist in determined orbitals around the nucleus. It has a melting point of 1064 degrees celsius. 79 electrons (white) successively occupy available electron shells (rings). 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. It has a melting point of 1064 degrees celsius... 2, 8, 18, 32, 18, 1.

79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. And for gold, each energy level will hold: The nucleus consists of 79 protons (red) and 118 neutrons (blue). Each orbital can hold a certain amount of electrons; 26/03/2020 · the gold atomic structure has 79 protons and 118 neutrons in its nucleus. Atomic structure of gold 1.1 atomic number. 79 electrons (white) successively occupy available electron shells (rings). 79 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Atomic structure of gold 1.1 atomic number.
